Unique Tips About How To Draw A Dipole Moment

The mostly used unit of dipole moment is debye (d).
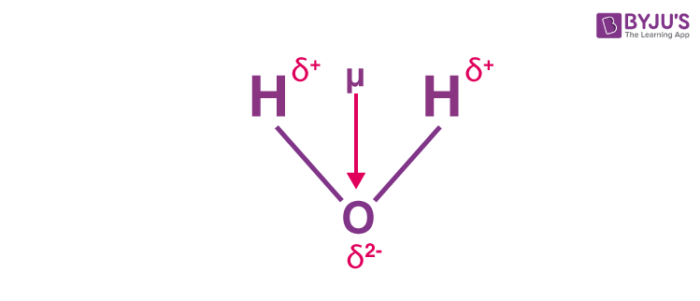
How to draw a dipole moment. Where, μ is the dipole moment vector. Q i is the magnitude of the i. It also explains how to calculate the percent.
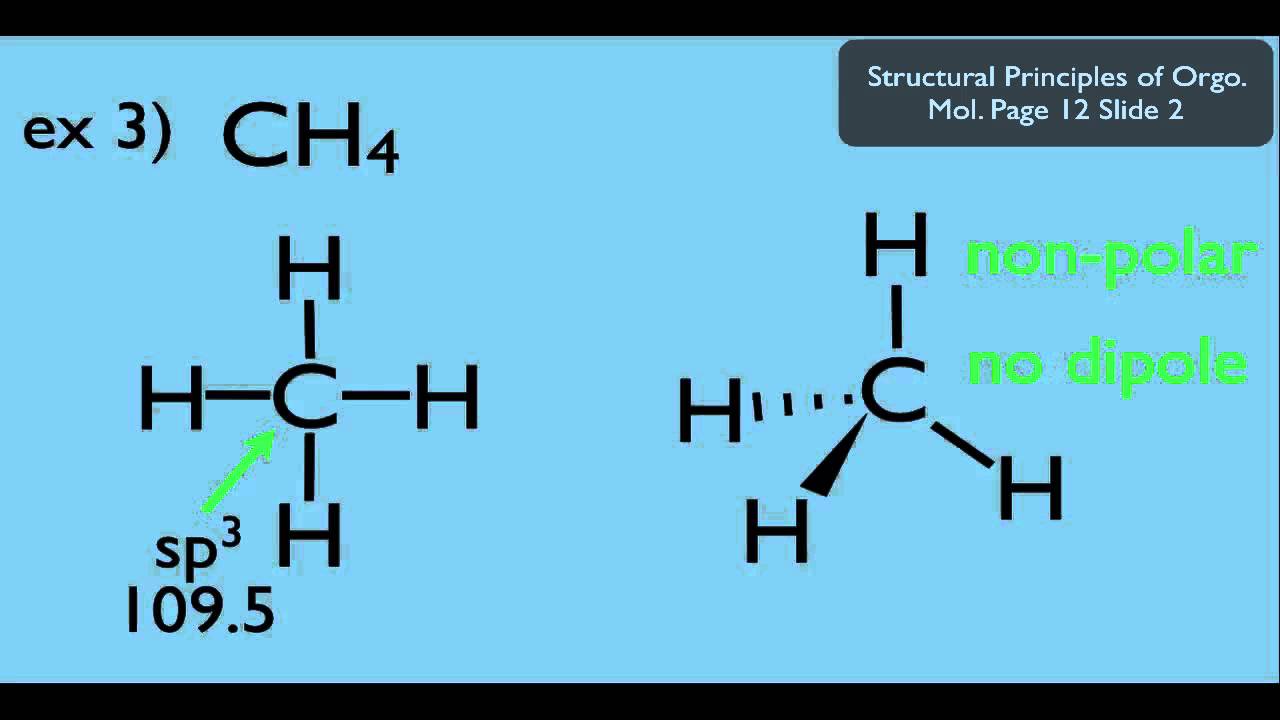
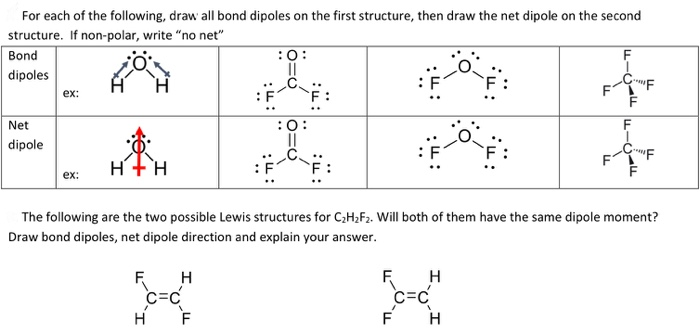
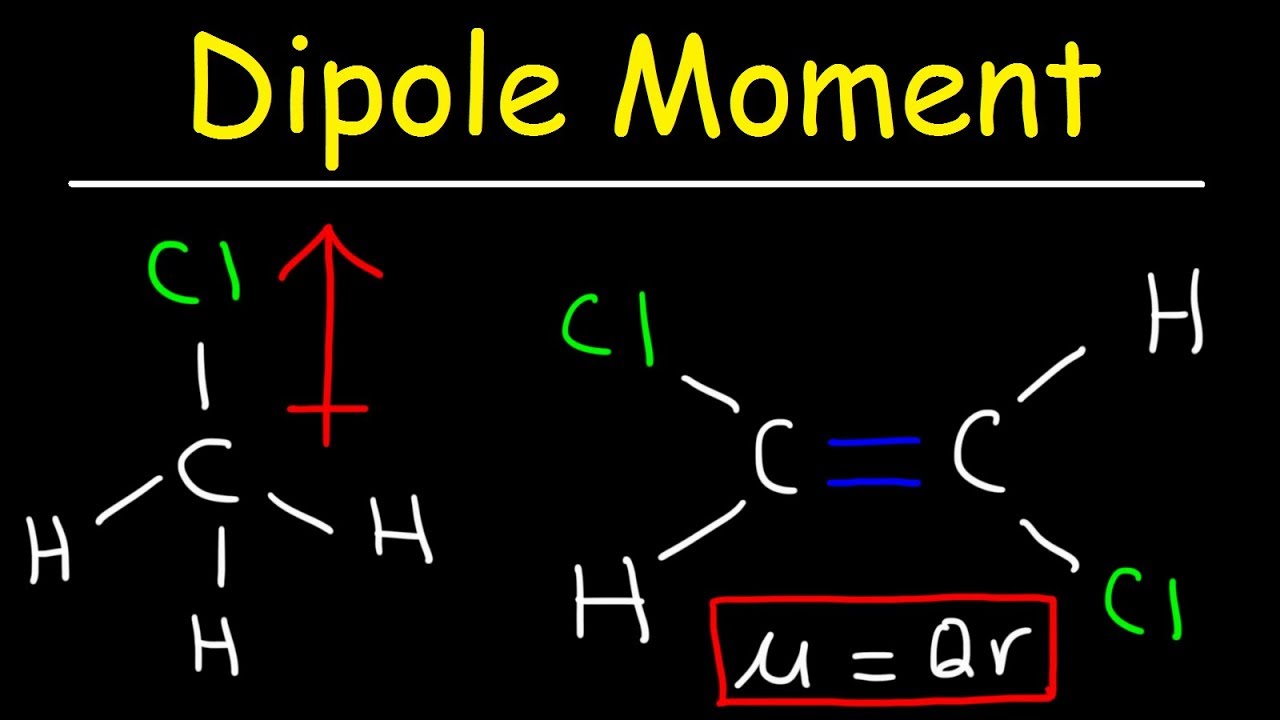
Here are some quick steps to determine the dipole moment of the molecule:step 1) obtain the lewis dot structure for the molecule. Is represented by the greek letter mu ( µ) and is given by the formula shown below, where q is the magnitude of the partial charges (determined by the electronegativity. Thus, the dipole moment, μ = q × d {\rm{\mu }} = q \times d μ = q × d;
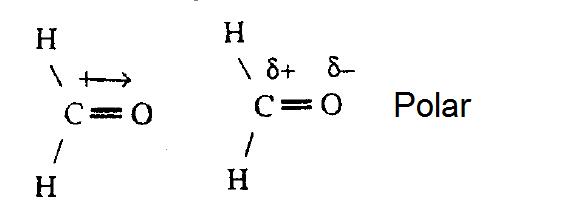
The delta positive pole is the atom that is lower in electronegativity. The dipole moment of a molecule can be calculated by another primary equation that is mentioned below: This organic chemistry video tutorial provides a basic introduction into dipole moment and molecular polarity.
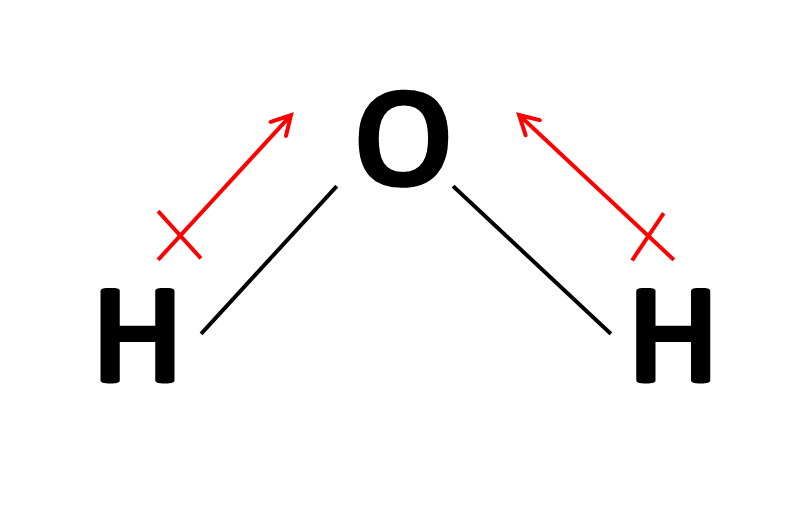
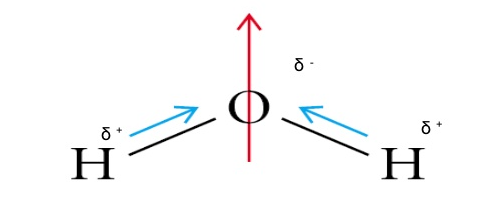
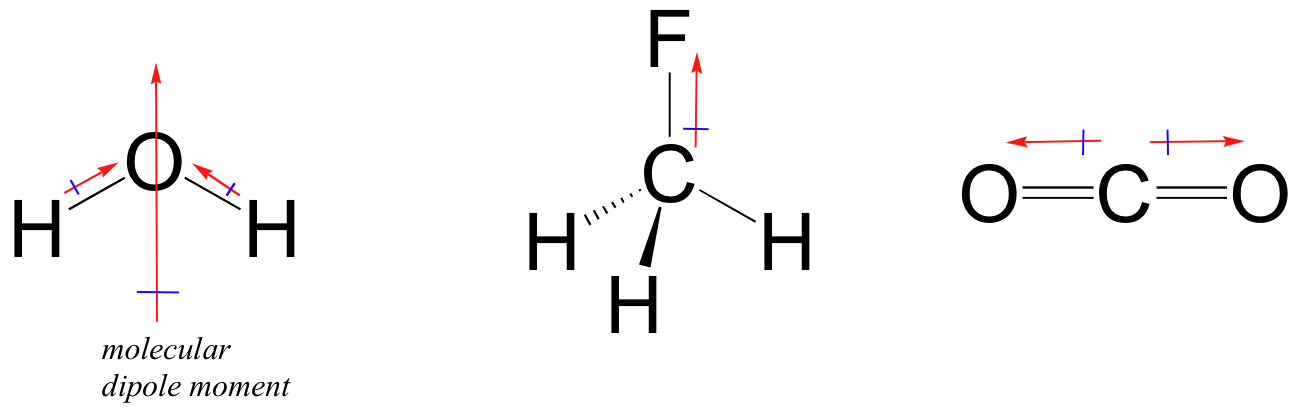
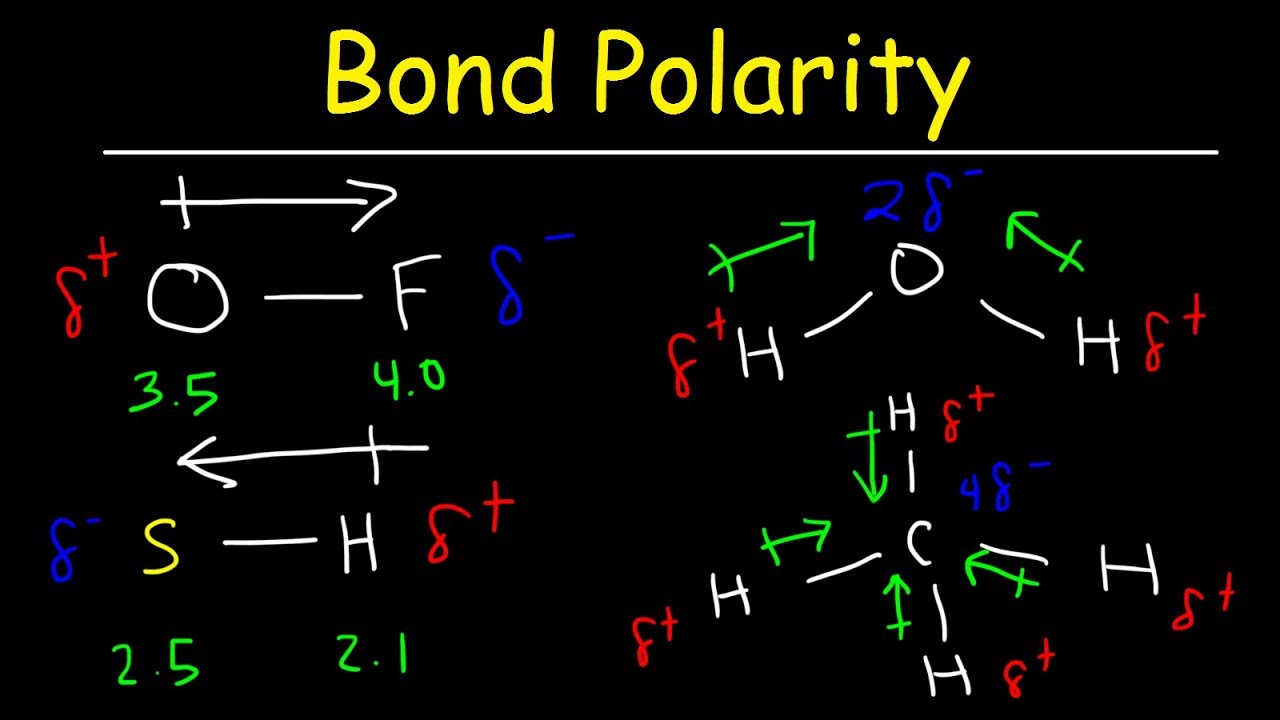
It explains how to indic. How to draw overall dipole moment november 25, 2021 a dipole moment is caused by the presence of one or more polar bonds ie bonds between two different elements. How do we draw the dipole moment of a water molecule?
The delta negative pole is the atom that is higher in electronegativity. Looking at the electronegativity and shape of the \(\mathrm{h}_{2} \mathrm{o}\) molecule tells you how the. The symbols have their significant meanings.
This chemistry video tutorial provides a basic introduction into bond polarity, electronegativity, and the dipole moment of a bond. Dipole moment in a lewis structure.